 The most critical issues in 14C dating are the sampling process, the sample size and the presence of various contaminants. wood, canvas, leather, vegetal fibres, etc. The wide-spread use of the method is mainly due to the ubiquity of the organic materials found in artwork, e.g. Radiocarbon (hereafter 14C) dating has become an important dating method in cultural heritage science, especially for authentication and forgery detection. 
The radiocarbon method was first developed for dating organic compounds in mid 1940s by the physical chemist Willard Frank Libby at the University of Chicago. The radiocarbon dating method and textiles The 14C date of textiles therefore reflects the time of production, but not necessarily the time of consumption or final use. However, in textiles the fibers are spun and woven into cloth, which could be used for decades or generations before the textile was deposited in a grave or discarded. This date gives the time of harvesting the plant fibres or collecting the animal fibres or filaments. Since 14C decays at a constant rate, calculations can be made to estimate when replenishment stopped, by measuring the residual amount of 14C remaining in the organic matter, thus enabling dating of an excavated find. Once simple plants (as opposed to woody trees) are cropped, animal hair is cut, and silk filaments are produced, replenishment of 14C stops. The heaviest carbon isotope, 14C is constantly created in the Earth’s atmosphere by neutrons which are secondary cosmic rays, subsequently concerted to carbon dioxide (CO 2), and thus incorporated into living organisms, like photosynthetic plants, and then to the rest of the biosphere. Carbon has three isotopes, they all have six protons, but each isotope has six ( 12C), seven ( 13C) or eight ( 14C) neutrons. Isotopes of an element have the same number of protons but different numbers of neutrons. on the number of protons and neutrons present. Since the matter of electrons is extremely small, the mass of an atom is based on the matter within the nucleus, i.e. Each atom has a nucleus, containing protons and neutrons, and a number of electrons orbiting around it. An isotope is one of two or more species of atoms of a chemical element with the same atomic number but different atomic masses. 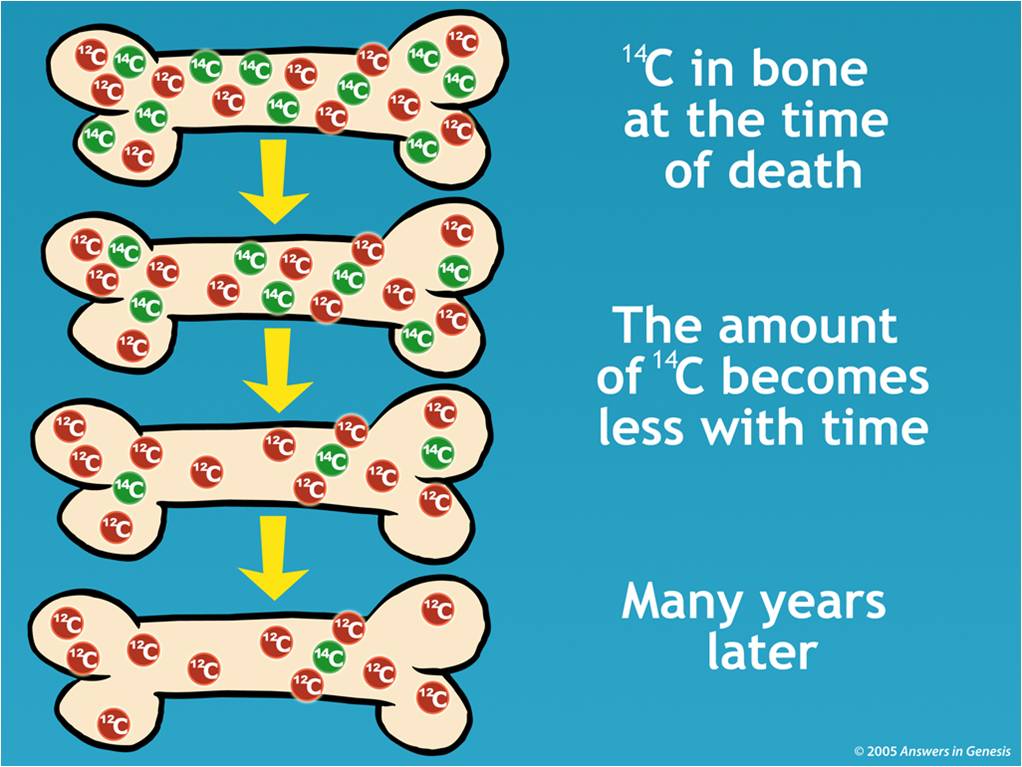
Radiocarbon dating, also referred to as carbon dating or carbon-14 dating ( 14C), is a method that can calculate the age of materials of plant or animal origin, by measuring the amount of the radioactive isotope of carbon (C), the 14C isotope, present in the material. The C of excavated textiles might also be affected at some point after excavation and during conservation treatment as their condition is often so poor it requires the use of synthetic adhesives or consolidants to retain their structure. The purpose of this paper is to present and discuss how 14C results might be affected by the condition of the textiles, when that condition has disturbed the C content of the material analysed or when provenance information does not exist to corroborate 14C results. 
This study shows that efficiency of the technique is highly dependable on the amount of carbon present in the finds, which does not seem to be affected by carbonisation, but is greatly affected by mineralisation and the incorporation of foreign organic matter, like synthetic consolidants.ġ4C dating is based on measurements of residual 14C within the organic material analysed. This paper includes a discussion and review of the dating method, focusing on sample selection, sample preparation, and by evaluating the efficiency of the technique on textile finds at various states of preservation. Two carbonised textile finds, two which were mineralised (one treated with adhesives in the past), and a final two which come from inhumation burials (similar to the textile previously treated with adhesives). Six case studies of archaeological textiles dated using the carbon-14 method are presented here. However, the quantity and quality of carbon present in the organic matter of archaeological textiles can be affected either by the conditions under which the find was preserved (such as carbonisation, chemical change of organic matter to carbon, and mineralisation, which is the gradual replacement of organic matter by metal degradation products), or by the application of certain interventive methods of conservation (such as, consolidation and the application of organic adhesives that add foreign organic matter/carbon to the textile). Radiocarbon dating provides objective age estimates of archaeological finds, based on measurements of the carbon-14 isotope present in the organic matter against an internationally used reference standard. Archaeological textiles are suitable material for radiocarbon dating as they are mainly made of organic matter, such as plant and/or animal fibres. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
